Toxoplasma gondii is a single-celled protozoan parasite responsible for toxoplasmosis infection and disease in humans. This parasite infects a majority of warm-blooded species and birds around the globe, with its primary host consisting of the Felidae family (domestic cat species and close relatives) [1].
There are three infectious stages/forms of T. gondii throughout the parasite’s lifecycle: tachyzoites (in groups or clones), the bradyzoites (in tissue cysts), and sporozoites (in oocysts) [2]. While all forms can result in Toxoplasma gondii infections if ingested (by cats and humans) the typical cycle begins with the interaction with oocysts found in the environment. From initial stages of infection the parasite can either enter a reproductive replication cycle within the host, or form latent tissue cysts which can persist throughout a lifetime (maturing through the various infectious stages of T. gondii). Re-activation (or shift from the cyst to active reproduction phase) can occur spontaneously or as a result of environmental factors, leading to renewed tachyzoite replication and possible oocyst shedding (in felines) [2] [1].
There are three infectious stages/forms of T. gondii throughout the parasite’s lifecycle: tachyzoites (in groups or clones), the bradyzoites (in tissue cysts), and sporozoites (in oocysts) [2]. While all forms can result in Toxoplasma gondii infections if ingested (by cats and humans) the typical cycle begins with the interaction with oocysts found in the environment. From initial stages of infection the parasite can either enter a reproductive replication cycle within the host, or form latent tissue cysts which can persist throughout a lifetime (maturing through the various infectious stages of T. gondii). Re-activation (or shift from the cyst to active reproduction phase) can occur spontaneously or as a result of environmental factors, leading to renewed tachyzoite replication and possible oocyst shedding (in felines) [2] [1].
Tachyzoites
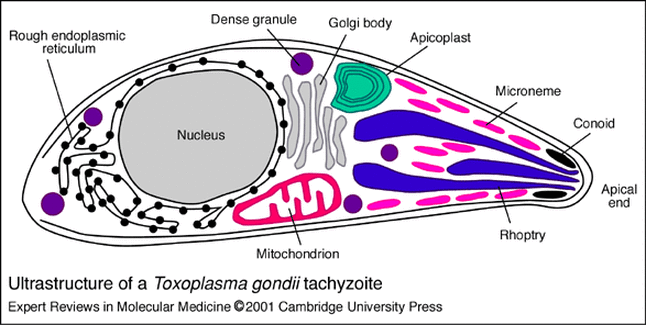
Figure 1 Structure of mature Tachyzoite of Toxoplasma gondii. Parasitic particles contain many of the same structures as metazoan cells including ER, Golgi, and Mitochondria. Additional structures such as the micronemes, conoids, and granules are typical in protozoa and contribute to motility, host cell invasion, and phagocytosis. [2]
Seen as the singular infectious parasites of T. gondii, tachyzoites are composed of the same organelles as many metazoan cells (golgi, nucleus, ER, etc.) with a longer, slender appearance. The parasite is typically 2 by 6µm in size and has a shape similar to that of a crescent with their anterior ends slightly pointed [2]. During the active (lytic) phase of T. gondii infections, the tachyzoites penetrate the host cellular membrane using their actin-dependent microneme structures [5], or enter the cell via phagocytosis wherein they multiply asexually (creating two new T. gondii tachyzoites). Once replication can no longer be facilitated within the host, the cell ruptures, releasing the newly formed parasitic units into the extracellular matrix/bloodstream [2].
|
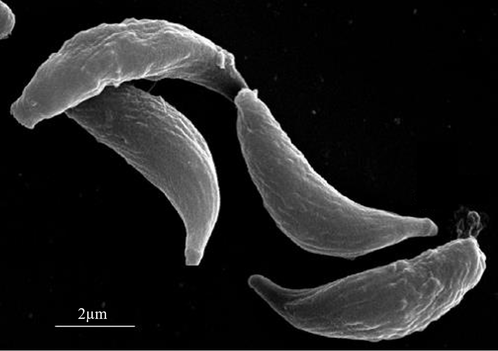
Figure 2 Scanning electron microscopy of T. gondii Tachyzoites. Parasites are typically 2µm wide and 6µm in length, with an overall crescent appearance. The anterior (conoidal) end of Toxoplasma gondii used for host cell entry and can be identified by its elongated shape; the posterior end is rounded in comparison. [2] |
Bradyzoites
|
During the chronic (latent) phase of Toxoplasma gondii infections, bradyzoites (also referred to as cystozoites) replicate slowly within infected tissue, forming tissue cysts from initial tachyzoite infection [1]. Depending on the site of colonization, tissue cysts may range in size from two to hundreds of bradyzoites, growing up to 100µm. Cysts formation occurs most commonly in neural, eye, and spinal tissue (these exhibit a smaller, circular appearance); however intramuscular bradyzoite cysts can occur and generally have an elongated, larger morphology [2]. The main observable differences between bradyzoites and tachyzoites in the T. gondii infectious cycle is a more slender appearance of tissue cyst bradyzoites; additional differences in tissue staining and composition can be observed through clinical/laboratory microscopy [2]. Unlike T. gondii tachyzoites, bradyzoite tissue cysts remain with the host throughout their lifetime, presenting the ability for re-activation of the parasite later down the line; in some cases, these tissue cysts can die off. Tissue cysts within muscle/neural cells continually replicate bradyzoites at low levels throughout their lifetime; rupturing of cysts (due to trauma, reactivation, etc.) can result in the release of parasites back into the body [2]. These parasites are typically quickly controlled by the host's immune system (preventing additional disease/infection), however those who are immunosuppressed such as AIDS patients may exhibit recurrence of disease symptoms [3]. Toxoplasmosis infections (symptomatic) seen in HIV/AIDS or other immunocompromised individuals are typically a result of bradyzoite (tissue cyst) re-activation rather than a new host-parasite interaction [3] [8]. |
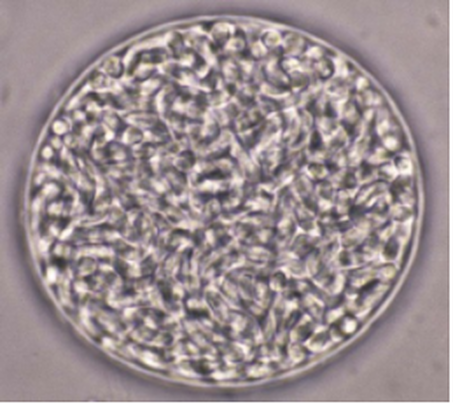
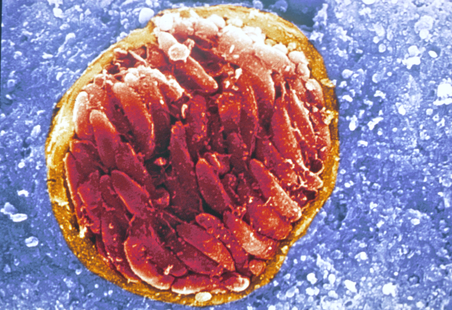
Figure 3 Brain tissue cyst containing bradyzoites in infected mice. Cysts caused by latent Toxoplasma gondii infections (after Tachyzoite phase) exhibit thin cell walls and persist in the host throughout their lifetime. Bradyzoites may form cysts throughout the body, mainly colonizing muscular and neural tissues. [2]
Figure 4 Toxoplasma gondii tissue cysts in neural tissue
|
Sporozoites
Sporozoites are the immature (non-infectious) particles of T. gondii infection which reside within sporocysts and oocysts produced in the digestive tracts of felines after approximately two days of asexual reproduction. Unlike tachyzoites which divide, replicate, and infect cells within the host actively, oocyst (sporozoite) formation results in the spread of toxoplasma gondii particles into the environment. When ready to form oocytes, T. gondii creates male and female gamonts; these structures are similar to that of the sperm/egg in animals where the smaller male gamont ‘swims’ to fertilize a large female gamont within the cells of the small intestine [2]. Once fertilized, an oocyte forms which eventually develops a singular unsporulated sporocyst.
|
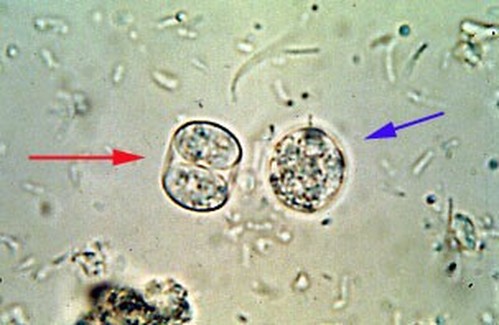
Figure 5 (Right) Toxoplasma gondii oocysts containing immature bradyzoites. Unsporulated cysts (blue) are shed through the digestive tract of cats where they mature into sporulated cysts (red) containing sporozoites. These sporulated cysts contain infectious bradyzoites which are then injected by a host; upon which they transform into infectious tachyzoite particles. [2]
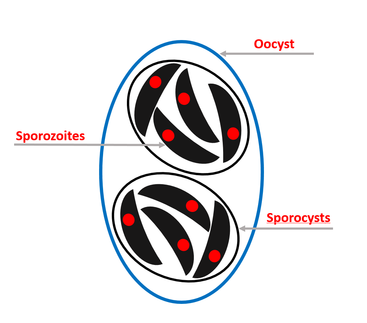
Figure 6 (Below) Sporulated T. gondii oocytes containing sporocytes and sporozoites. Once mature, oocysts contain two separate sporocysts, each in turn containing four sporozoites (immature Toxoplasma gondii particles). [2] |
|
The term unsporulated refers to the presence of an undivided sporocyst which lacks mature infectious particles [2]. Sporulation (maturation) then occurs within the oocyst (often once its secreted by a cat), resulting in the formation of two sporocysts; each of these structures then go on to form four individual T. gondii parasites (for a total of eight) (Figure 6) which can then further mature into tachyzoites [1].
|
Life Cycle
The Toxoplasma gondii infectious cycle can begin at a variety of different points in the parasites developmental cycle. The only known definitive host for the parasite, in which it can undergo both sexual reproduction ad asexual replication, are domestic (and some wild breeds) of cats [1].
|
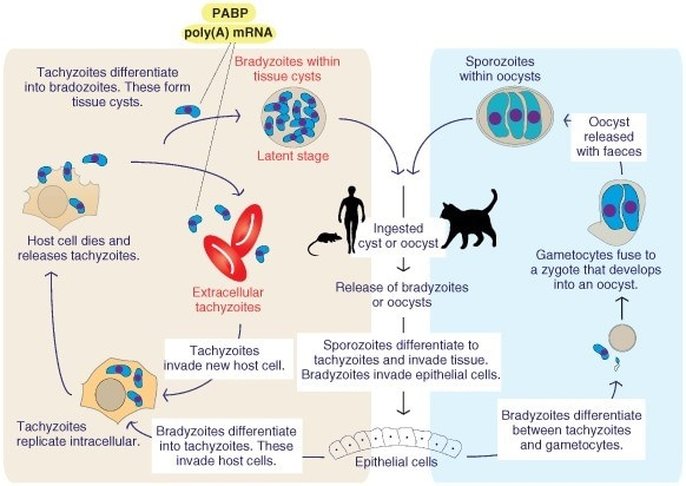
Figure 7 Toxoplasma gondii infection begins through ingestion of tissue cysts or oocysts within the environment. Parasites differentiate into infectious tachyzoites which invade host cells, bradyzoites invade epithelial cells within the digestive system of the host. In an established host species (felines) bradyzoites in the intestine undergo sexual reproduction in the form of gametocytes, these come together to form oocysts which are excreted by the host (sporozoites). In non-feline infections, tachyzoites replicate within host cells resulting in their destruction. Eventual immune response leads to maturation of the tachyzoites into latent bradyzoites, leading to muscular/neural tissue cysts. [2] [8]
|
Cats may become infected through ingestion of intermediate hosts containing T. gondii tissue cysts; additionally direct contact with oocysts (shed from infected cats) can confer infection. Once ingested, the parasites travel through the digestive system wherein the cell wall of the sporocyst or tissue cysts is dissolved, releasing the infectious particles [8]. After entry into the host cells, the parasite begins to replicate as tachyzoites, which then spread throughout the body of the host, establishing infections mainly in neural and muscle tissue. As the feline immune system begins to gain immunity to the parasite, the number of extracellular tachyzoites drops and the formation of tissue cysts (bradyzoites) occurs [8]. During this infectious period, T. gondii undergoes sexual reproduction using gametocytes; oocysts develop and are excreted through the digestive system into the environment for an average of 1-2 week following infection [8].
|
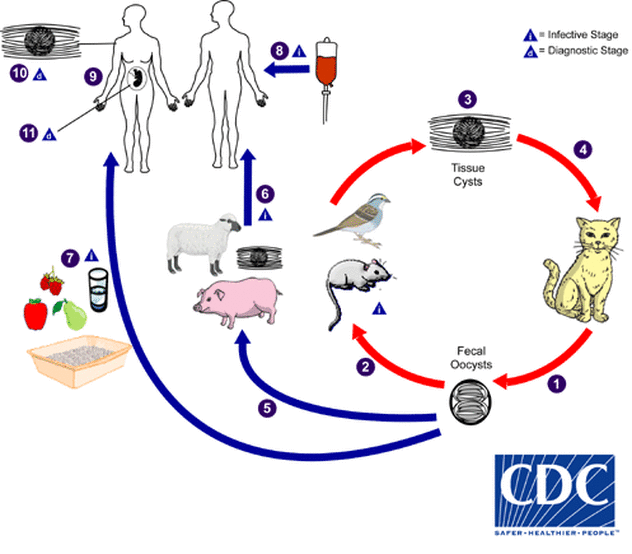
Figure 8 Oocysts shed via infected felines (1) are ingested by secondary hosts such as birds, pigs, and mice (5)(2). Tissue cysts containing latent parasites are either re-ingested by cats, wherein T. gondii undergoes replication and sexual reproduction (4) or are consumed by other mammals. Human infection can result from ingestion of tissue cysts from livestock (undercooks meat) (6) or from contact with infectious oocysts in the environment/on fomites (7). Infection results in the eventual formation of tissue cysts (containing the parasite) which remain with a person throughout their lifetime. Additionally, T. gondii may be passed through infected blood donations (8) or contracted congenitally when a mother is infected during pregnancy (9). Congenital toxoplasmosis can lead to various health problems in the developing fetus, including hydrocephalus, developmental issues, and deafness/blindness (11) [1].
|
Once excreted into the environment, oocysts sporulate or mature (forming two sporocysts with four sporozoites each) within an average of 1-5 days under ideal climate/environmental conditions. In this state, the oocysts are highly stable and can remain infectious for long periods of time depending on the temperature/moisture levels [8]. Those shed into the soil may confer infection for up to a year, while those in cool water can remain infectious for several years [8]. Humans (or intermediate hosts such as other mammals) become infected through either ingestion of sporulated oocysts or tissue cysts form infected meat. Once introduced to the human body, T. gondii undergoes a typical 5-23-day incubation period wherein disease/symptoms are not yet observable [8]. After the initial incubation period, Toxoplasma gondii infections progress through the same developmental steps/phases as in their definitive hosts, cats. Tachyzoites invade and replicate quickly within muscle/neural cells until the hosts immune system begins to develop resistence, wherein bradyzoites establish tissue cysts throughout various tissues in the body [1]. Tissue cysts last throughout the lifetime of an infected person, and have the potential of re-activating infection if the host environment (immune system) becomes compromised [1]. The exception to this parallel infection is the fact that humans (and other mammals) do not see a sexual reproduction of the parasite during T. gondii infection, resulting in no shed oocysts. In this way, infection cannot be passed between persons via sporozoites; this leads to the ‘dead-end host’ situation in which Toxoplasma gondii can only complete its lifecycle through a feline host and ‘stays put’ in other, intermediate species.